Stem Cells, What they are, How to use them and their future implications
Imagine if you could cure many diseases such as AIDS, Diabetes, Stroke and Alzheimer with just one element, would you be astonished by that? What would happen if I told you that this element already exists and they are called stem cells?

What’s a stem cell?
A stem cell is an unspecialised cell that must be able to be self-renewing and be multipotent. Self-renewing means that the cell must be able to replicate itself into more cells of the same unspecialised cell type. In other words, stem cells can make copies of themselves over and over again.
This term is different from Multipotent cells. Multipotent cells are unspecialised cells and must be able to differentiate into many specialised cell types. These cell types compose the tissues and the organs of an individual.
Even if the DNA is always the same, any cell is different from each other for structure or function. That depends on the tissue or the organs.
An old saying goes: “We aren’t all equal”. In a certain way, it is the same with stem cells. There are a variety of different stem cells used in research. Stem cells differ through various times during human development.
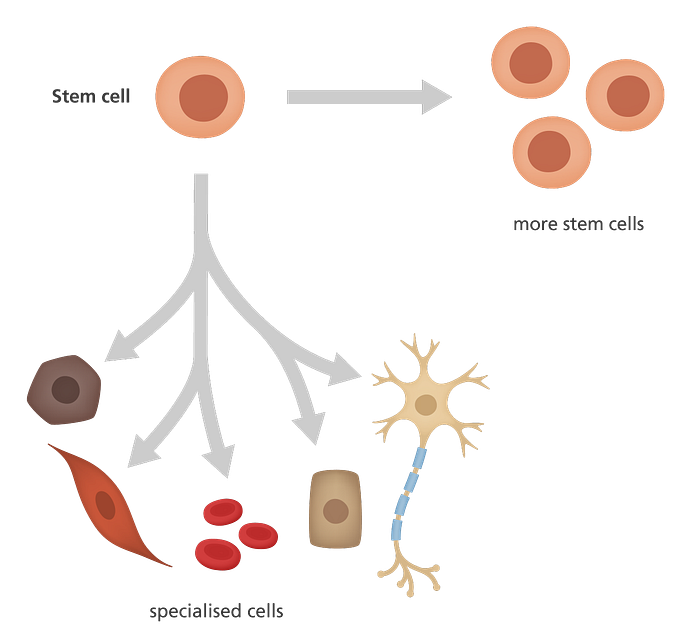
We have 3 types of stem cells:
- Embryonic stem cells
- Cord Blood stem cells
- Adult Stem Cells
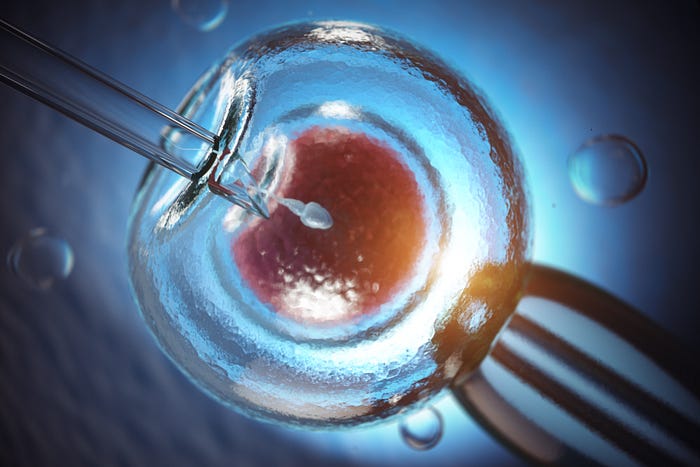
Embryonic stem cells are a product of In vitro fertilisation. They are made in a petri dish after that eggs are fertilised, and then they develop into blastocysts.
Cord Blood stem cells are the cells that come from umbilical cords just after a baby is born.
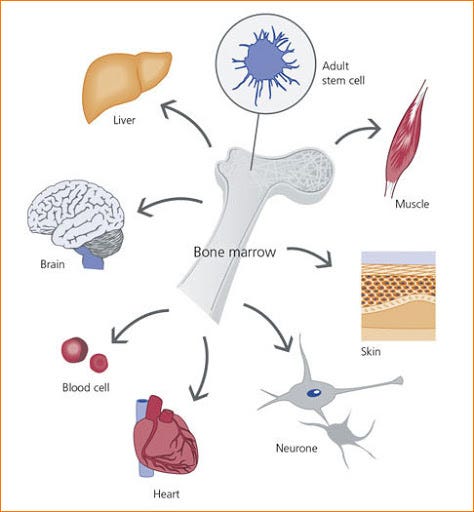
Adult stem cells are found in the body of an adult, and they are present in all of us. Scientists have found adult stem cells in the brain, in bone marrows, in the blood, in blood vessels, in skeletal muscles, in the skin and the liver.
The story of stem cells began in the 17th century with the discovery of the microscope. A Dutch father-son team named Hans and Zacharias Janssen invented the first so-called compound microscope in the late 16th century when they discovered that, if they put a lens at the top and bottom of a tube and looked through it, objects on the other end became magnified. Even if now it could be something ordinary, at that time it was like magic.
In 1665, an English scientist and architect, recently called “England’s Leonardo”, Robert Hooke, using a microscope, became the first person in the world to visualise a microorganism. Hooke’s discovery led to a whole in the field of scientific study known as cell biology. It may seem obvious to us now, but it took scientists another 200 years after Hooke’s discovery to confirm that all living things are made of cells.
Just after that, Schleiden collected data from different plants and Schwann collected cells from animals; they were stunned to see that they were all made up of cells. They are the authors of what is called “Cell Theory”
In 1855, Rudolf Virchow, the father of modern pathology, proposed what was called the Biogenic Law: “Omnis cellula e cellula”, or “All living cells arise from pre-existing cells”. Today, we know that this process is one cell division in which a single cell divides to make two copies of the same cell.
What makes stem cells so special is that they don’t just replicate themselves, they also can become multiple different kinds of cells. The discovery of that phenomenon was the contribution of physicians and scientists together. While cell biologists were studying cells, doctors were looking for ways to save their patients from excessive bleeding.
The first successful human blood transfusion was performed at the beginning of the 19th century by Dr. James Blundell in England. He was able to save a patient who suffered excessive blood loss during childbirth by transferring blood from her husband to her. She was lucky, but not all blood transfusions were successful. This raised questions about what Scientists and physicians wanted to know, what determines whether a blood transfusion was accepted or rejected? More interestingly, for the future of stem cell research, scientists began to wonder, what was in the fresh blood that helped patients recover?
In the early 20th century a Nobel prize-winning German physician and scientist, Paul Ehrlich, had developed staining techniques that made it possible to distinguish between different types of blood cells under a microscope.
In 1905, Artur Pappenheim produced a cell fate diagram suggesting that a specific cell type could give rise to all the other blood cell types. He proposed the term “stem cell” to define all different blood cell types.
At the same time, Theodor Boveri and Valentin Häcker used the term stem cell to describe cells committed to give rise to the germline. Scientists also realised that stem cells were abundant in the bone marrow. Bone marrow is a spongy tissue found inside of some of our bones such as hip, thigh bones and at the proximal ends of the long bones femur and humerus.
It took another 50 years to prove the existence of blood stem cells by experiments. In the late 1950s, Doctor McCulloch and Till in Canada began to explore how radiation affects human cells. They studied that because The Cold War was in full swing at that time, and they were seeking ways to protect soldiers from the side effects of radiation. They knew that massive radiation depleted the blood cells and caused genetic abnormalities at a molecular level. For this reason, they tried to inject bone marrow cells exposed to different levels of radiation. The highly radiated ones (because they were dead from the amount of exposure) didn’t give rise to any lumps or nodules. However, bone marrow cells exposed to low levels of radiation gave rise to nodules. When they looked at those nodules, they discovered that transplanted bone marrow cells were replicating themselves and giving rise to multiple cell types.

But were they replicating and differentiating from one single cell? Yes.
All the cells in the nodules had the same unique genetic abnormality. This new understanding marked the discovery of the first adult stem cells. After having discovered stem cells in bone marrow, scientists started finding new places to find them. In some organs such as the gut and skin, scientists discovered stem cells that regularly divide to repair and replace worn out or damaged tissue. An example is what happens when we replace dead skin cells with new ones. In other organs, however, such as the brain and heart, stem cells only divide under particular conditions.
Another type of stem cells is umbilical cord blood cells. These cells are known to be another good source of hematopoietic, also known as blood stem cells. The main features are that Cord blood cells are less invasive to collect, and these cells are naive that the cell transplantation recipients show less immune reactivity in the case of cell transplantations.
Some people may decide that, when they have a baby, the blood is collected from the placenta and umbilical cord under sterile conditions to isolate and freeze them for long term storage. In this way, children could potentially have a way to cure themselves in the future.
During all this situation, other 2 scientists, Doctors Edwards and Patrik Steptoe in England were trying to help patients who had fertility problems. They attempted to mature and fertilise a woman’s egg outside of the body in a Petri dish, a process known as in vitro fertilisation. In 1978, they figured out the culture conditions and were able to get early-stage embryos, and blastocyst in a culture dish, which they then successfully transferred to a mother. The result was the world’s first test-tube baby, Louise Brown. She was the first human to have been born after conception by in vitro fertilisation. She has been called the World’s first test-tube even if her conception took place in a Petri dish.
In 1998, Jamie Thompson, an American Developmental Biologist, used this technology to isolate the cell clumps from embryos leftover from in vitro fertilisation and generated the first human embryonic stem cell line. He was able to expand these cells for a long time without losing their capacity for self-renewal. They were 220 cell types, divided into 3 embryonic germ layers: ectoderm, mesoderm and endoderm.
What is the difference between these cells?
To answer this question, we should consider what Potency is.
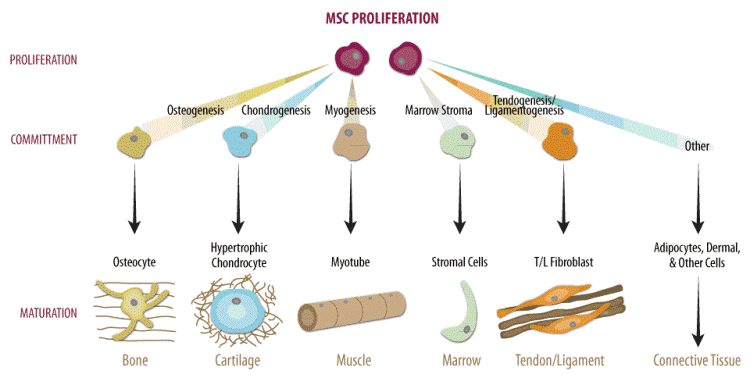
The potency of cells refers to a cell’s ability to make different cell types and germ layers. It comes from the Latin word potent, which means having power. The higher the potency, the higher the number of cell types a cell can differentiate into. Embryonic stem cells are pluripotent because they can become any type of cell of the body.
Quoting the example made by Dr. Zehra Dincer, if you’re a pluripotent stem cell, you are on the top of a mountain, while you are skiing, you can take many different weights to go down, assuming each path makes another cell type. As pluripotent stem cells, you can differentiate into all the cell types of the body. If you’re a multipotent cell, you’re starting lower down than the pluripotent cell on the potency mountain, such as adult stem cells. You can still pick many ways to go down. However, you can’t go up skiing, so you can only differentiate into a handful of cell types.
Even if there are these 3 types of stem cells, there is a fourth important one that was discovered by Shinya Yamanaka in Japan. As a child in Osaka, Yamanaka enjoyed taking clocks and radios apart and putting them back together. After beginning his career as a surgeon, he was so much frustrated about not being able to heal some of the worst diseases on Earth.
Yamanaka started wondering whether he could reprogram regular adult cells, like skin cells, to become stem cell-like cells. This concept can turn cells pluripotent again. After years of painstaking research, in 2006, Yamanaka and his team discovered that proteins encoded by just four “master genes” could turn any adult cell back into a stem cell. He called these cells Induced Pluripotent Stem Cells or IPSCs. Suppose we want to use the analogy of the mountain, we can go on the top of the hill, again.
Using own stem cells is so important because if people have to use “allogenic stem cells” (stem cells that are not present in your body), the body would not be able to recognise those cells. With Allogenic stem cells, there could be a risk of rejection, and you may require immunosuppression that can have many risks and many other problems.

Stem cells can provide multiple applications:
The study of early human development
Stem cell research benefits the study of development stages that cannot be studied directly in a human embryo. These developmental stages are linked with significant clinical consequences such as congenital disabilities, pregnancy loss and infertility.
The study of diseases in a culture dish outside of the body
Pluripotent stem cells, the genomes that predispose the disease, can be used to develop cellular models of human diseases. Scientists can test if a mutation is responsible for a disease. In this way, scientists don’t need to do experiments on actual patients. In other words, you could directly test something without harnessing anybody.
Based disease models can be used for screening new drug candidates that can slow or prevent diseases understanding their different response to drugs.
Before the stem cell revolution, people used to test therapies just on patients. Stem cells can help scientists to figure out the potential side effects of these existing drugs. This problem is present because scientists only test drugs for a specific organ without considering the entire situation. By using human stem cell drive, liver, kidney, brain, and pancreas cells, the new drugs can be tested in a culture dish first for its side effects rather than testing in human subjects. It will accelerate the drug discovery process and lead to safer and more effective treatments. For the application of stem cells, our stem cell therapies, as you have seen stem cells, have the potential to make every kind of cell in the body.
They can provide benefits in the field of therapeutic cloning and regenerative medicine.
We could stem cells to replace or restore damaged tissues such as diabetes, heart attacks, Parkinson disease, skin burns, or a spinal cord injury. The mission, here, is to find creative ways of delivering stem cells into injury sites. Another opportunity could be to fight ageing because stem cells could restore damaged tissues.

“Technology is neither good nor bad; nor is it neutral.”
Stem cells could have multiple problems
The possible problem of immoral actions
The use of embryonic stem cells for research involves the destruction of blastocysts formed from laboratory-fertilized human eggs. For those people who believe that life begins at conception, the blastocyst is a human life and to destroy it is immoral and unacceptable.
We don’t know long-term effects
Like any other new technology, it is also completely unknown what the long-term effects of such an interference with nature could materialise.
You can’t completely control stem cells
You can’t control what a stem cell will become when it’s inside your body. Many unauthorised therapies, for instance, may cause many problems for your health.

Potential future implications:
What if you could regrow an amputated hand or regenerate damaged brain cells? Some animals, such as salamanders, cockroaches, or seastars, can regrow their limbs. One question we should ask is
“If they can, why can’t we?”
Mammals, in fact, have a limited ability to regenerate their tissues. One aspect of regeneration that appears to be common to most organisms is the use of stem cells to replace missing tissues.
Axolotls or Mexican salamanders, for example, have extensive regenerative abilities. Not only can they regenerate limbs, but they can also regenerate their eyes, spinal cord, jaw, tail, and skin without any scarring. You can also even cut a part of their spine, in a month and a half, and it will have grown back. You would not be able to tell that you cut a piece of their spine out in the first place. After the spine is cut, the cells on the side of the cut lose their identity. So instead of being skin cells or blood cells, they become pluripotent stem cells and they can make any cellular tissue that the body will need to repair itself. In this case, axolotls use the pluripotent stem cells to regenerate all the cells to create a new and perfect spine. This phenomenon is a favourite among the scientific community because of its potential to help amputees, burn victims, and cancer patients. The study of regeneration helps scientists to learn how adult tissues heal and rebuild themselves. Researchers are interested in whether we can potentially use this information for human patients in a clinical setting.
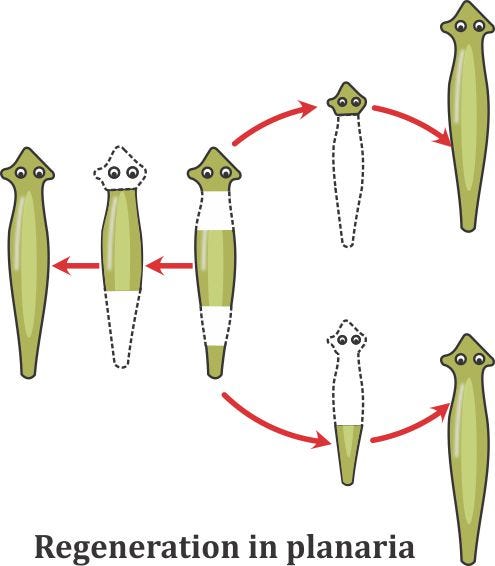
This example isn’t equal to another group of organisms called planarians, which are flatworms. If you cut them in hundreds of pieces, any of them will regrow to form a new example of the same species.
These abilities aren’t just related to animals; even plants have them. Rock cress is a small flowering plant related to cabbage and mustard. They have a different type of stem cells called Meristems. Meristem comes from the Greek word Merist(os), which means divide or division. Rock cress’s stem cells are located in two areas. The first one is in their growing tips where they help the length of the plant grow. They make new leaves, flowers, and stems.
The second one is Root tips meristems grow the roots so they can reach more resources from the soil.

To sum up, we have considered:
- the four types of stem cells,
- the history behind stem cells,
- the term potency,
- why stem cells are so important
- what problems can be caused and
In any case, we can be sure that stem cells are the cellular beginnings of a radical transformation in our world
